Ethylene Sulfate: Uses, Properties and Safety Guide
Ethylene sulfate is an important organic compound widely used in chemical and industrial applications. It belongs to the class of sulfate esters and is derived from ethylene glycol. Due to its reactive nature, ethylene sulfate plays a crucial role in synthesis processes and manufacturing industries. Understanding its properties, uses, and safety aspects is essential for both researchers and professionals working in chemistry-related fields.
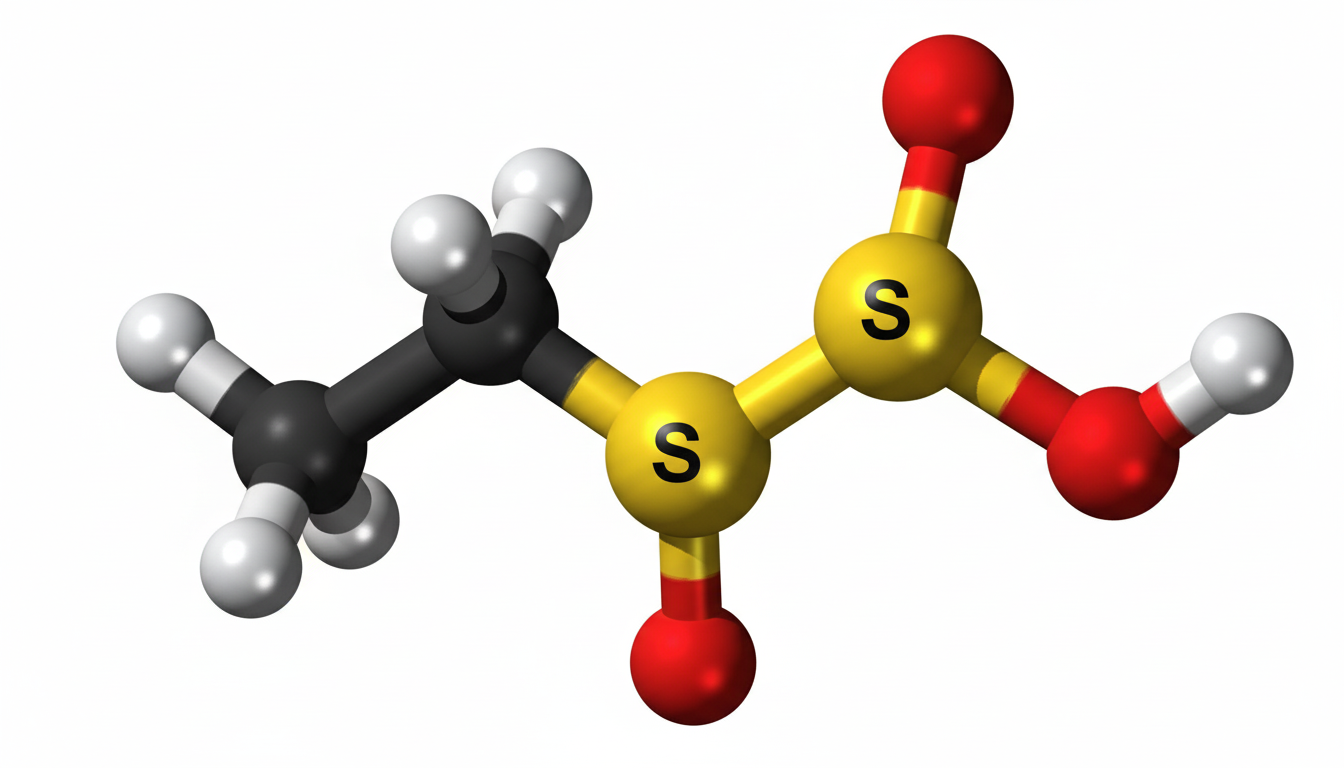
Chemical Structure and Composition
Ethylene sulfate has a cyclic structure that includes a sulfate group bonded to an ethylene backbone. This structure gives it unique chemical properties, making it useful in various reactions. It is typically represented as a cyclic sulfate ester formed from ethylene glycol and sulfuric acid derivatives. The compound is known for its stability under controlled conditions but can react under specific environments, especially in the presence of nucleophiles.
Physical Properties of Ethylene Sulfate
Ethylene sulfate appears as a colorless to pale yellow liquid under standard conditions. It has a relatively high boiling point compared to many organic compounds, which makes it suitable for industrial processes that require heat stability. The compound is soluble in polar solvents such as water and alcohols, allowing it to be used in various chemical formulations. Its density and viscosity also contribute to its usability in manufacturing environments.
Chemical Properties and Reactivity
One of the most important characteristics of ethylene sulfate is its reactivity. It can undergo ring-opening reactions when exposed to nucleophiles, making it a valuable intermediate in organic synthesis. This property is particularly useful in producing polymers, pharmaceuticals, and specialty chemicals. Additionally, ethylene sulfate can hydrolyze in the presence of water, leading to the formation of ethylene glycol and sulfate ions.
Industrial Applications of Ethylene Sulfate
Ethylene sulfate is widely used in the chemical industry due to its versatility. It serves as an intermediate in the production of polymers and resins. Many industries use it in the synthesis of surfactants and detergents, where its chemical properties enhance product performance. It is also utilized in laboratories for research purposes, especially in reactions requiring sulfate ester intermediates.
Role in Polymer and Plastic Production
In polymer chemistry, ethylene sulfate is used as a building block for creating advanced materials. Its ability to react with other compounds allows manufacturers to produce polymers with improved strength, flexibility, and durability. These materials are commonly used in packaging, construction, and automotive industries. The compound’s role in enhancing polymer properties makes it highly valuable in modern manufacturing.
Use in Pharmaceutical and Chemical Research
Ethylene sulfate is also used in pharmaceutical research for synthesizing complex molecules. Scientists use it as a reagent to introduce sulfate groups into organic compounds. This process is essential in drug development and biochemical studies. Its controlled reactivity allows researchers to design specific reactions and achieve desired outcomes in laboratory settings.
Safety and Handling Precautions
Handling ethylene sulfate requires proper safety measures due to its reactive and potentially hazardous nature. It can cause irritation to the skin, eyes, and respiratory system if not handled correctly. Protective equipment such as gloves, goggles, and lab coats should always be used. Proper ventilation is also necessary when working with this compound to avoid inhalation of vapors.
Health Hazards and Toxicity
Exposure to ethylene sulfate may pose health risks, especially with prolonged or high-level contact. It can be harmful if inhaled, ingested, or absorbed through the skin. Symptoms of exposure may include irritation, discomfort, and in severe cases, chemical burns. Therefore, it is important to follow safety guidelines and use the compound only in controlled environments.
Environmental Impact of Ethylene Sulfate
Ethylene sulfate can have an environmental impact if released into water or soil. Due to its chemical nature, it may affect aquatic life and disrupt ecosystems. Proper disposal methods should be followed to minimize environmental risks. Industries are encouraged to implement waste management practices that reduce the release of harmful chemicals into the environment.
Storage and Transportation Guidelines
To maintain its stability and safety, ethylene sulfate should be stored in tightly sealed containers away from moisture and heat. It should be kept in a cool, dry place with proper labeling. During transportation, it must be handled according to chemical safety regulations to prevent leaks or accidents. Using approved containers and following guidelines ensures safe handling throughout its lifecycle.
Advantages of Using Ethylene Sulfate
Ethylene sulfate offers several advantages in industrial and research applications. Its high reactivity allows for efficient chemical synthesis, reducing production time and cost. It is also versatile, making it suitable for a wide range of uses. Additionally, its stability under controlled conditions makes it reliable for manufacturing processes.
Limitations and Challenges
Despite its benefits, ethylene sulfate has certain limitations. Its reactive nature can pose safety risks if not handled properly. It may also require specific storage conditions to maintain stability. Furthermore, its environmental impact and toxicity require careful management, which can increase operational costs for industries.
Future Prospects and Research Developments
Research on ethylene sulfate continues to explore new applications and safer alternatives. Scientists are working on developing eco-friendly processes that reduce its environmental impact. Innovations in chemical engineering may lead to improved methods of using this compound in sustainable ways. Its role in advanced materials and pharmaceuticals is expected to grow in the future.
Conclusion
Ethylene sulfate is a valuable chemical compound with a wide range of industrial and research applications. Its unique properties make it essential in polymer production, chemical synthesis, and scientific studies. However, its use requires careful handling and proper safety measures to minimize risks. By understanding its characteristics and applications, industries and researchers can utilize ethylene sulfate effectively while ensuring safety and environmental protection.
- Art
- Causes
- Best Offers
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Games
- Festival
- Gardening
- Health
- Home
- Literature
- Music
- Networking
- Other
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness



